Weathering and Soils
Weathering and Soils
back to Contents of Entire Course...
Physical Weathering
Chemical Weathering
Types of Chemical
Weathering Reactions
Weathering of Common Rocks
Weathering Rinds,
Exfoliation, and Spheroidal Weathering
Factors that Influence
Weathering
Soils
Soil Erosion
adapted to HTML from lecture notes of Prof. Stephen A. Nelson Tulane
UniversityWeathering is the process that breaks rocks down to smaller fragments and alters minerals formed at higher temperature and pressure to those stable under conditions present near the Earth's surface.
Geologists recognize two categories of weathering processes
- Physical Weathering - disintegration of rocks and minerals by a physical or mechanical process.
- Chemical Weathering - chemical alteration or decomposition of rocks and minerals.
Physical Weathering
Physical weathering takes place by a variety of processes. Among them are:
- Development of Joints - Joints are regularly spaced fractures or cracks in rocks that show no offset across the fracture (fractures that show an offset are called faults).
- Joints form as a result of expansion due to cooling or relief of pressure as overlying rocks are removed by erosion.
- Joints form free space in rock by which other agents of chemical or physical weathering can enter.
- Crystal Growth - As water percolates through fractures and pore spaces it may contain ions that precipitate to form crystals. As these crystals grow they may exert an outward force the can expand or weaken rocks.
- Heat - Although daily heating and cooling of rocks do not seem to have an effect, sudden exposure to high temperature, such as in a forest or grass fire may cause expansion and eventual breakage of rock. Campfire example.
- Plant and Animal Activities -
- Plant roots can extend into fractures and grow, causing expansion of the fracture. Growth of plants can break rock - look at the sidewalks of New Orleans for an example.
- Animals burrowing or moving through cracks can break rock.
- Frost Wedging - Upon freezing, there is an increase in the volume of the water (that's why we use antifreeze in auto engines or why the pipes break in New Orleans during the rare freeze). As the water freezes it expands and exerts a force on its surroundings. Frost wedging is more prevalent at high altitudes where there may be many freeze-thaw cycles.
Chemical Weathering
Since many rocks and minerals are formed under conditions present deep within the Earth, when they arrive near the surface as a result of uplift and erosion, they encounter conditions very different from those under which they originally formed. Among the conditions present near the Earth's surface that are different from those deep within the Earth are:
- Lower Temperature (Near the surface T = 0-50oC)
- Lower Pressure (Near the surface P = 1 - several hundred atmospheres)
- Higher free water (there is lots of liquid water near the surface, compared with deep in the Earth)
- Higher free oxygen (although O is the most abundant element in the crust, most of it is tied up bonded into silicate and oxide minerals, at the surface there is much more free oxygen, particularly in the atmosphere).
Because of these different conditions, minerals in rocks react with their new environment to produce new minerals that are stable under conditions near the surface. Minerals that are stable under P, T, H2O, and O2 conditions near the surface are, in order of most stable to least stable:
- Iron oxides, Aluminum oxides - such as hematite Fe2O3, and gibbsite Al(OH)3.
- Quartz*
- Clay Minerals
- Muscovite*
- Alkali Feldspar*
- Biotite*
- Amphiboles*
- Pyroxenes*
- Ca-rich plagioclase*
- Olivine*
The main agent responsible for chemical weathering reactions is water and weak acids formed in water
- An acid is solution that has abundant free H+ ions.
- The most common weak acid that occurs in surface waters is carbonic acid.

Types of Chemical Weathering Reactions
- Hydrolysis - H+ or OH- replaces an ion in the mineral.

- Leaching - ions are removed by dissolution into water. In the example above we say that the K+ ion was leached.
- Oxidation - Since free oxygen (O2) is more common near the Earth's surface, it may react with minerals to change the oxidation state of an ion. This is more common in Fe (iron) bearing minerals, since Fe can have several oxidation states, Fe, Fe+2, Fe+3. Deep in the Earth the most common oxidation state of Fe is Fe+2.
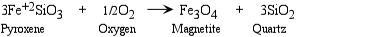
- Dehydration - removal of H2O or OH- ion from a mineral.
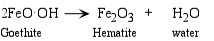
- Complete Dissolution - all of the mineral is completely dissolved by the water.
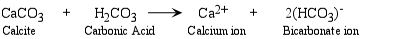
Weathering of Common Rocks
| Rock | Primary Minerals | Residual Minerals* | Leached Ions |
|---|---|---|---|
| Granite | Feldspars | Clay Minerals | Na+, K+ |
| Micas | Clay Minerals | K+ | |
| Quartz | Quartz | --- | |
| Fe-Mg Minerals | Clay Minerals + Hematite + Goethite | Mg+2 | |
| Basalt | Feldspars | Clay Minerals | Na+, Ca+2 |
| Fe-Mg Minerals | Clay Minerals | Mg+2 | |
| Magnetite | Hematite, Goethite | --- | |
| Limestone | Calcite | None | Ca+2, CO3-2 |
*Residual Minerals = Minerals stable at the Earth's surface and left in the rock after weathering.
Weathering Rinds, Exfoliation, and Spheroidal Weathering
When rock weathers, it usually does so by working inward from a surface that is exposed to the weathering process. This may result in:
- Weathering Rinds - a rock may show an outer weathered zone and an inner unweathered zone in the initial stages of weathering. The outer zone is known as a weathering rind. As weathering continues the thickness of the weathering rind increases, and thus can sometimes be used as an indicator of the amount of time the rock has been exposed to the weathering process.
- Exfoliation - Concentrated shells of weathering may form on the outside of a rock and may become separated from the rock. These thin shells of weathered rock are separated by stresses that result from changes in volume of the minerals that occur as a result of the formation of new minerals.
- Spheroidal Weathering - If joints and fractures in rock beneath the surface form a 3-dimensional network, the rock will be broken into cube like pieces separated by the fractures. Water can penetrate more easily along these fractures, and each of the cube-like pieces will begin to weather inward. The rate of weathering will be greatest along the corners of each cube, followed by the edges, and finally the faces of the cubes. As a result the cube will weather into a spherical shape, with unweathered rock in the center and weathered rock toward the outside. Such progression of weathering is referred to as spheroidal weathering
Factors that Influence Weathering
- Rock Type
- Different rocks are composed of different minerals, and each mineral has a different susceptibility to weathering. For example a sandstone consisting only of quartz is already composed of a mineral that is very stable on the Earth's surface, and will not weather at all in comparison to limestone, composed entirely of calcite, which will eventually dissolve completely in a wet climate.
- Bedding planes, joints, and fractures, all provide pathways for the entry of water. A rock with lots of these features will weather more rapidly than a massive rock containing no bedding planes, joints, or fractures.
- If there are large contrasts in the susceptibility to weathering within a large body of rock, the more susceptible parts of the rock will weather faster than the more resistant portions of the rock. This will result in differential weathering.
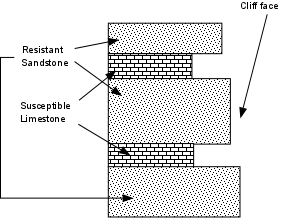
- Slope - On steep slopes weathering products may be quickly washed away by rains. On gentle slopes the weathering products accumulate. On gentle slopes water may stay in contact with rock for longer periods of time, and thus result in higher weathering rates.
- Climate- High amounts of water and higher temperatures generally cause chemical reactions to run faster. Thus warm humid climates generally have more highly weathered rock, and rates of weathering are higher than in cold dry climates. Example: limestones in a dry desert climate are very resistant to weathering, but limestones in a tropical climate weather very rapidly.
- Animals- burrowing organisms like rodents, earthworms, & ants, bring material to the surface were it can be exposed to the agents of weathering.
- Time - since a rate is how fast something occurs in a given amount of time, time is a crucial factor in weathering. Depending on the factors above, rates of weathering can vary between rapid and extremely slow, thus the time it takes for weathering to occur and the volume of rock affected in a given time will depend on slope, climate, and animals.
Soils
Soils are an important natural resource. They represent the interface between the lithosphere and the biosphere - as soils provide nutrients for plants. Soils consist of weathered rock plus organic material that comes from decaying plants and animals. The same factors that control weathering control soil formation with the exception, that soils also requires the input of organic material as some form of Carbon.
When a soil develops on a rock, a soil profile develops as shown below. These different layers are not the same as beds formed by sedimentation, instead each of the horizons forms and grows in place by weathering and the addition of organic material from decaying plants and plant roots.
- Caliche - Calcium Carbonate (Calcite) that forms in arid soils in the K-horizon by chemical precipitation of calcite. The Ca and Carbonate ions are dissolved from the upper soil horizons and precipitated at the K-horizon. In arid climates the amount of water passing through the soil horizons is not enough to completely dissolve this caliche, and as result the thickness of the layer may increase with time.
- Laterites - In humid tropical climates intense weathering involving leaching occurs, leaving behind a soil rich in Fe and Al oxides, and giving the soil a deep red color. This extremely leached soil is called a laterite.
- Paleosols - If a soil is buried rapidly, for example by a volcanic eruption, the soil may be preserved in the geologic record as an ancient soil called a paleosol.
Soil Erosion
In most climates it takes between 80 and 400 years to form about one centimeter of topsoil (an organic and nutrient rich soil suitable for agriculture). Thus soil that is eroded by poor farming practices is essentially lost and cannot be replaced in a reasonable amount of time. This could become a critical factor in controlling world population.

