Control of Air Pollution
Control of Air Pollution
adapted to HTML from lecture notes of Prof. Stephen A. Nelson Tulane UniversityControl of Air Pollution Introduction
Smog
Air Inversion
Global Warming
Greenhouse Gases
Acid Rain
Ozone Depletion (.gif animation)
Sources
see also Acid Rain page...
Control of Air Pollution
Automobiles Catlytic converter
- Converts carbon monoxide, nitrous oxides and hydrocarbons into carbon dioxide, nitrogen and water.
Recirculating exhaust gas along with a catlytic converter greatly reduces emissions.
Coal
Main problem is the sulfur dioxide it produces upon burning. Solutions: Burn only low sulfur coal.
Wash coal to remove pyrite.
Convert coal to gas (called Gasification)
Fluidized Bed Combustion (or Limestone Injection): Mix coal with limestone, to form calcium sulfides or sulfates, which are prevent sulfur from forming sulfur dioxide.
Scrubbing - slurries of lime or limestone.
Smog
Smoke plus Fog (U.K.) (Grey smog). Photochemical Smog - brown air.
(U.S.A.) Results from exhausts from cars, buses and trucks.
Nitrogen Oxide + Oxgen + Hydrocarbons + Sunlight -- > Ozone
6ppm ozone is deadly - We need ozone in stratosphere; at lower altitudes, however, it can kill.
Sulfurous Fog- gray air.Dominantly from burning coal with a significant pyrite content. The fogs of London in the past were actually sulfuous fog. This type of smog is no longer common in London as a result of environmental regulations on burning coal.
Air Inversion
In valleys or on the lee side of mountains, air inversion may occur. A
warmer air mass moves above cooler air, trapping the cooler, denser air
underneath and increasing the severity of air pollution. Los Angeles is a
good example of this, where warm desert air from the east comes over the
mountains to the east of Los Angeles and lies over the cooler Pacific
Ocean air. The cooler air is trapped because it cannot rise through the
less dense warm air above it, and the pollution in the cold air
accumulates.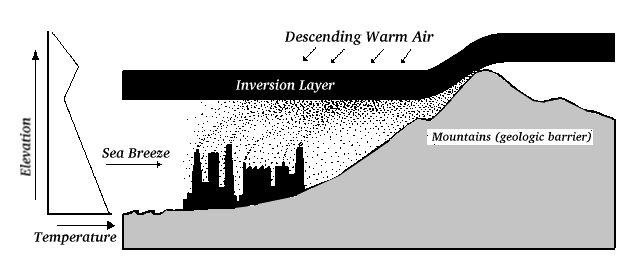
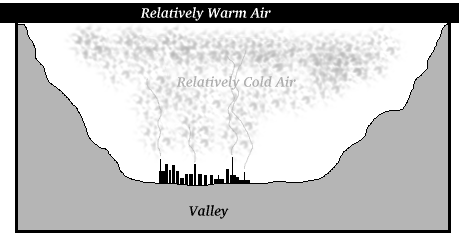
A similar situation arises in mountain valleys where warm air overlies the colder air which accumulates in the valleys.
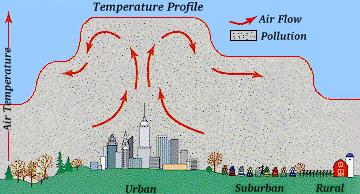
Also, cities tend to form featuers known as heat islands or dust domes, which tend to collect warm air filled with pollutants, and help spread it out over nearby suburbs.
Global Warming
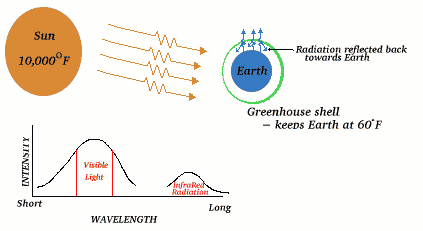
If there were no greenhouse effect, the average temperature on the earth would be - 20C with huge fluctuations in a 24 hour period. When visible light from the sun hits the earth some is absorbed, heating the earth. The heated earth gives off infrared radiation characteristic of the 25C temperature. This longer wavelength infrared radiation is then reradiated back into space. Some of the infrared radiation, however, is trapped (absorbed) by greenhouse gases.
This is known as The Greenhouse Effect.
There are several gases which cause the greenhouse effect, and whose concentrations determine the amount of heat retained by the earth. Primary among these are water vapor, carbon-dioxide and methane. The chart below shows how the concentration of CO2 in the atmosphere has changed in the last 40 years:
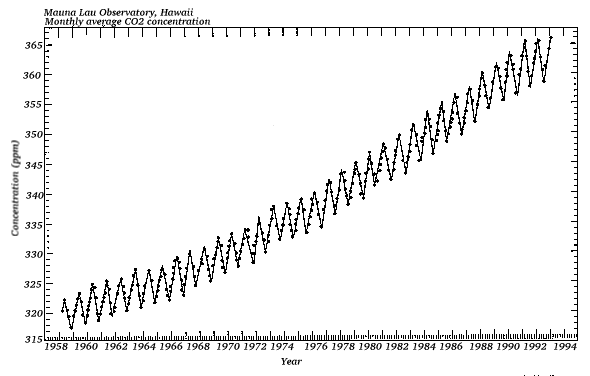
The increase in CO2 is anthropogenic (related to human activities). It is mostly due to the burning of fossil fuels and deforestation.
| Gases contributing to Anthropogenic Greenhouse Effect | ||
|---|---|---|
| Gas | Rate of Increase (% per year) |
Relative Contribution(%) |
| CO2 | 0.5 | 60 |
| CH4 | 1 | 15 |
| N2O | 0.2 | 5 |
| O3 | 0.5 | 8 |
| CFC-11 | 4 | 4 |
| CFC-12 | 4 | 8 |
Global Warming has been studied extensively, and currently, a large percentage of the scientific community have reached a consensus on various issues related to global warming. They are listed below:
| Scientific Consensus on Global Warming | |
|---|---|
| Statement | Consensus |
| Fundamental Physics | 90+% |
| Added greenhouse gases add heat | 90+% |
| Added gases are anthropogenic | 90+% |
| Reduction of uncertainty will require a decade | 90+% |
| Full recovery will require many centuries | 90+% |
| Large stratospheric cooling | 90+% |
| Precipitation will increase | 90% |
| Reduction of sea ice | 90% |
| Warming in arctic | 90% |
| Rise in sea level | 90% |
| Local details of global change | ? |
| Tropical storms increase | ? |
| Details of next 25 years | ? |
Greenhouse Gases
Carbon Dioxide (CO2): Roughly 60% of total
Burning Fossil Fuels
Deforestation
Methane (CH4): 15% of total
Coal Mines
- Termites
- Wetlands
- Rice Paddies
- Cattle
- Subpolar Soil and Wetlands (Methane Hydrate)
Acid Rain
Main source of acid rain is sulfur dioxide and nitrogen oxides in the atmosphere.
Power plants are a major source for sulfur dioxide and as well nitrogen oxide.
Automobiles, trucks and buses are major contributors of nitrogen oxides in urban and suburban environments.
Effects of Acid Rain
- If the bedrock consists of limestone, effect is not so severe. The limestone helps to neutralize the acid. If bedrock is granitic, there is no buffer Results severe.
- Acid solutions free nutrients as well as toxic metals from the soil. As a result, nutrients are lost from the soils and the plants may take on toxic elements.
- In lakes, acidity keeps the nutrients in solution. Nutrient are then lost with the outflow of water. Algae cannot grow as a result, and so there is no food for aquatic animals present in the lake.
- Fish may be poisoned by heavy metals in solution, and are very intolerant of high acidity.
- High acidity kills trees.
- Tree ring studies have shown that concentrations of calcium (which is an essential nutrient for trees) have been decreasing steadily in areas of increased acid rain.
- Acidic rain damages buildings and monuments.
Ozone Depletion

Sources
Acid Rain
- The Environmental Protection Agency acid rain page: http://www.epa.gov/docs/acidrain/
- A good general guide to acid rain and acid rain resoures online: http://qlink.queensu.ca/~4lrm4/
- A somewhat more technical page on acid rain: http://www.igc.org/acidrain/
Air Pollution
- Clean Air Act: http://gurukul.ucc.american.edu/TED/CLEAN.HTM
- A collection of Links on Air Pollution: http://www.deb.uminho.pt/fontes/enviroinfo/air.htm
Global Warming
- NASA Climate News page: http://www.climatenews.com/
- The Union of Concerned Scientists have a global warming page: http://www.ucsusa.org/warming/index.html
- The Sierra Club page: http://www.toowarm.org/home.html
- The Environmental Protection Agency site on Global Warming: http://www.epa.gov/globalwarming/
- This is a page arguing against global warming: http://www.webaxs.net/~noel/gwarming.htm
- This page details the myths and misarguments by both sides of the global warming debate: http://members.aol.com/jimn469897/warming.htm
- A Collection of sites that NASA put together about global warming: http://gcmd.gsfc.nasa.gov/faq/globwarm.html

